The first battery was a “voltaic pile”. A more improved battery in 1836 was the Daniel Cell. A battery is something that consists of at least one electrochemical cell, and that stores energy to be used as electricity. Daniell cell was was made in 1836, improved over other batteries.
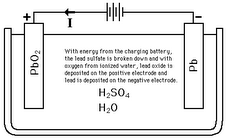
Batteries work by converting chemical energy to electrical energy. One half-cell has electrolyte in it, and an electrode to which negatively charged ions go to. The other half of the cell includes electrolyte and the electrode to which positively charged ions go to. In the redox reaction that powers the battery, electrons get added at the end of the positive electrode, while the electrons are removed at the anode.
There are two main types of batteries: Primary and Secondary batteries. Primary batteries are commonly referred to as “disposable batteries” and secondary batteries are referred to as “rechargeable batteries”. Further inquiry reveals that there are more subsets of lead-acid batteries (Which fall under secondary), such as Automotive, Flooded Cells, AGM and Gel Cells. Automotive batteries are only for short-term usage and should not be used off a grid. Flooded, or wet cells, are very common lead-acid batteries that are not sealed, thus the user can check and replenish the electrolyte. Has a lower cost. AGM batteries use something like fibreglass as a separator to hold the electrolyte in place. AGM batteries are spill proof and impact resistant. Sealed and cannot be refilled with electrolyte, do not overcharge. Uses same voltage set points as flooded cells, can be used as replacement for them. Other types of non-lead acid batteries include Zinc-Carbon (Primary), Zinc-Chloride (Primary), Alkaline (Primary, very common, also known as Zinc-Manganese dioxide), Nickel Cadmium (Secondary), and NiMH (Secondary).
An electric current is a flow of electric charge. Electric charge flows when there is voltage present across a conductor.
A circuit is a roughly circular line, route or movement that starts and finishes at the same place. It works by flowing through wires, and the components are the battery, the switch, the ammeter, and light.
We are using a starting lead acid battery to power our boat. This means that it has thinner plates than a deep cycle battery, as well as it has more immediate power.